During the Dragon Boat Festival, the 2024NCCLab was held in Wuhan. Many experts conducted academic exchanges around clinical and laboratory testing and clinical quality system construction. I went to multiple conference rooms. They were all full and everyone was working hard.
There were many IVD companies exhibiting at the conference, and the most attractive thing was the new industry booth, which was well located, cleverly designed, and displayed rich content.


At the scene, I had some exchanges with Mr. Cheng Ran, the head of the new industry marketing department, and gained a deeper understanding of the logic of doing things in the new industry and observations of the future of the industry. In addition, I visited the new industry headquarters last year, and I gained a deeper understanding.
At the scene, New Industry demonstrated a series of software and hardware strength sectors such as the pre- and post-processing modules and ultra-high-speed tracks of the T8 assembly line, ixLab intelligent management system, quality control product series, standardized laboratory construction plans, industry-university-research cooperation, etc. Overall, it reflects the new industry’s continuous innovation and development concept around clinical problem solving, namely:
A new doctor.
New is innovation and a new industry.
Medicine is a clinical need and the core of high-quality development.
Mr. Cheng said that we continue to be oriented by clinical needs, increase technological innovation and academic investment, help comprehensively improve testing service capabilities, promote high-quality development of medical and health services, and contribute new industrial strength to the implementation of the "Healthy China 2030" strategy.
As a leading domestic luminescent IVD company, the impression given by the new industry is:
Steady and far-reaching progress.
Take one step at a time to achieve the ultimate in luminous products; strategically build smart laboratory solutions for the future.
The new industry has maintained a compound growth rate of over 22% in the past five years. In overseas markets, the five-year compound growth rate has reached 40%. In the capital market, the market value of new industries has maintained its first position in the IVD track for a long time.
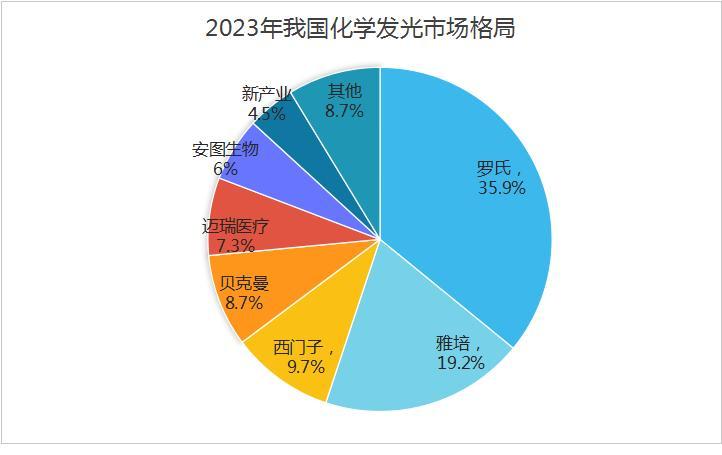
Biochemistry and immunity account for nearly 70% of clinical tests, and chemiluminescence is a promising growth track. The new industry is not in a hurry to expand its product line, but is taking a slow and steady approach, first maximizing its core biopharmaceutical products.
In terms of products, it has formed a continuously evolving series of high-speed light-emitting devices such as X3, X6, X8, and X10, which also have high cost-effective advantages.

In terms of reagent items, there are currently 175 chemiluminescence reagents registered in the domestic market. The supporting reagents cover tumor markers, thyroid, infectious diseases, gonads, myocardial markers, inflammation monitoring, glucose metabolism, bone metabolism, liver fibrosis, autoimmune antibodies and other testing items, ranking first in the country. There are 61 biochemical reagents, and the supporting reagents cover testing items such as kidney function, liver function, special proteins, lipids and so on.
In overseas markets, the company sells 199 chemiluminescent reagents globally, 188 chemiluminescent reagents approved by the EU, and 61 biochemical reagents.

Due to revolutionary technological breakthroughs in the field of small molecules, the new industry has introduced to the market a total of 6 reagents: 25-hydroxyvitamin D, aldosterone, estradiol, free triiodothyronine, total triiodothyronine, and tacrolimus, which solve the common problems of low sensitivity and poor accuracy in the current use of competitive methods to detect small molecules, making the products have better specificity, sensitivity, precision, and linearity.
In the latest announcement, the new industry's coagulation and molecular products have been laid out. The self-developed T8 assembly line has been launched at this year's CACLP, which will form a fully automatic immunobiochemical molecular coagulation analysis assembly line in the future.
In terms of full laboratory management, the new industry has also formed an intelligent and standardized management system for clinical testing, which can complete automatic quality control, automatic audit, and full-process management of instruments and reagents. It is also equipped with multi-disciplinary top-level analysis systems, worry-free services, ISO15189 services, and 6S services to put the concept of full laboratory solutions into practice.
I said,
There is no ceiling for the development of new industries.
Mr. Cheng said,
Because of continuous innovation, there is no ceiling.
The development of new industries in the past 30 years has been a journey of innovation, deeply fulfilling the slogan of bravely shouldering national responsibilities and adhering to technological innovation.
29 years ago, Dr. Rao Wei, who was born in technology, realized that Chinese companies must make breakthroughs in core technologies, otherwise it will only be a matter of time before they get stuck. In line with the responsibility and mission of "breaking technology monopoly and mastering core technologies" and "allowing Chinese people to spend less money on medical treatment and testing", Dr. Rao Wei established the first R&D team and started the independent research and development road "starting from a beaker". Since then, he has entered the no man's land of chemiluminescence technology where no Chinese are present.
In 2010, New Industry Biotech successfully developed and launched China's first fully automatic chemiluminescent immunoassay analyzer, breaking the monopoly of overseas giants on China's chemiluminescent immunodiagnostic market.
Subsequently, Dr. Rao Wei led the new industrial biological research and development technology team to quickly move to another no-man's land – independent research and development and production of key antigen and antibody raw materials.
After the key raw material R&D platform was established, the R&D team actively engaged in researching small molecule detection technology.
At present, the new industry has established four major technology platforms: nanomagnetic microsphere R&D, reagent key raw material R&D, diagnostic instrument R&D, and diagnostic reagent R&D, which will continue to assist the company in instrument R&D, reagent raw material R&D, and reagent R&D.
As a chairman with a technical background, Rao Wei has been managing research and development for many years, leading New Industrial Biotech to become a leading domestic chemiluminescent immunoassay company and moving towards a global pioneer in the field of in vitro diagnostics.
Nowadays, the new industry has entered the stage of R&D and innovation oriented to clinical needs, practicing the concept of "product development oriented to solving clinical problems" and starting to develop projects from the source.
Come for living water from a source.
In terms of industry, academia and research, new industries have built a smooth transformation channel around clinical needs, allowing the company to have a steady stream of new projects in the future.

In terms of cutting-edge research, we focus on scientific research cooperation to assist clinical diagnosis and treatment. Multi-center research on multiple topics has been carried out in conjunction with scientific research institutes and hospital terminals, such as the GADA-liver cancer early screening model, liver disease development prediction model, new standards for original aldehyde screening and diagnosis, high-sensitivity troponin reference interval for healthy people, etc.
In terms of innovative learning, we have established chronic disease management forums, new industry and new forums, and international clinical laboratory exchange conferences to promote clinical and laboratory academic exchanges.
In terms of product introduction, new detection products based on the small molecule sandwich method are constantly being developed; new indicators of bone metabolism are developed; four new coagulation indicators for early warning indicators of thrombotic diseases are launched.
Embrace centralized purchasing and accelerate domestic substitution.
Currently, centralized procurement is covering all areas of IVD, which is more beneficial to domestic leading companies with leading brands, rich products, and high market shares.
The new industry is exactly this type of enterprise, which has gained a lot in the process of embracing centralized purchasing.
In Anhui's first centralized procurement of luminescent reagents in 2021, 14 new industry projects won bids, accelerating the installation of luminescent high-speed machines and breakthroughs into high-end hospitals.
By the end of 2023, the centralized purchasing alliance organized by 25 provinces (districts and corps) including Anhui Province will cover eight testing items for infectious diseases and six testing items for sex hormones. All chemiluminescence detection projects covered by the new industry's centralized procurement have entered Group A and were successfully selected. The implementation will be promoted in 2024, which is bound to bring more breakthroughs in hospital installations.
In the wave of domestic substitution, new industries are the main force. In the domestic immunoluminescence market, 70% of the market share is still occupied by large imported brands. Looking to the future, this part of the market is bound to be gradually transferred to domestic representative companies. This is also the direction that national IVD brands need to work together. Innovative products, reliable quality, and high-quality services are needed, and new industries shoulder important responsibilities.
Yixin turned to medicine and rushed to the future.