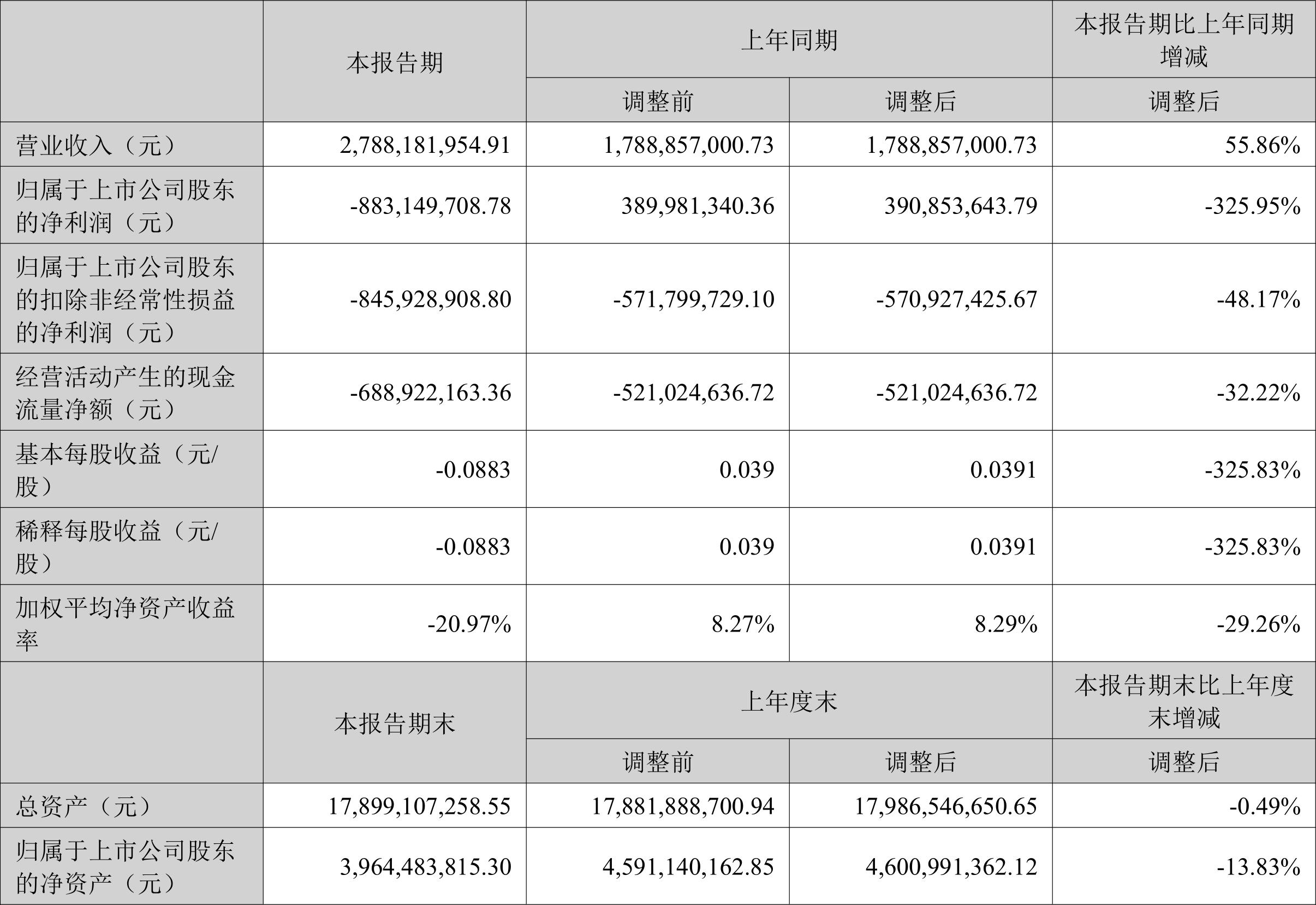
On March 24, Maiwei Biotech announced its 2025 annual report. The company's operating income was 663 million yuan, a year-on-year increase of 231.6%; the net profit attributable to the parent company increased from a loss of 1.04 billion yuan in the same period last year to a loss of 969 million yuan, and the loss decreased; Net profit after deducting non-attributed profits to the parent company changed from a loss of 1.07 billion yuan in the same period last year to a loss of 999 million yuan, and the loss decreased; net operating cash flow was -290 million yuan, a year-on-year increase of 69.7%; EPS (fully diluted) was -2.4258 yuan.
In the fourth quarter, the company's operating income was 96.68 million yuan, a year-on-year increase of 64.7%; the net profit attributable to the parent company increased from a loss of 350 million yuan in the same period last year to a loss of 372 million yuan, and the loss further expanded; the net profit after deducting non-attributable parent profits increased from a loss of 355 million yuan in the same period last year to a loss of 376 million yuan, and the loss further expanded; EPS was -0.9301 yuan.
As of the end of the fourth quarter, the company's total assets were 4.556 billion yuan, an increase of 6.6% from the end of the previous year; net assets attributable to the parent company were 350 million yuan, a decrease of 77.7% from the end of the previous year.
The company mentioned in its 2025 annual report that its business operations have undergone major changes in many aspects. As an innovative biopharmaceutical company, the company continues to promote the research and development of new drugs, and its overall R&D investment remains at a high level. During the reporting period, R&D investment was 977 million yuan, an increase of 24.79% compared to the same period last year. As of the disclosure date of this report, the company has 14 key varieties in preclinical research, clinical research or marketing stages, including 10 innovative drugs and 4 biosimilar drugs, focusing on tumors and age-related diseases.
In terms of product progress, the company has multiple innovative drugs in the critical registration clinical trial stage. In 2025, the company's first innovative drug, agostimin α for injection (Melansan®), was approved for marketing, further strengthening the company's commercial layout. At the same time, the company actively promotes clinical research on the varieties under development, including 9MW2821, 9MW1911, 7MW3711, etc., showing good clinical prospects. The company is also promoting product registration and market layout in many countries and regions, especially in emerging markets, and has made certain progress.
(Maiwei Biological Announcement)