The recommendation level of CSCO (Chinese Society of Clinical Oncology) guidelines is a bridge between high-level medical evidence and complex clinical decision-making. It is not just a simple grading, but a comprehensive consideration of the strength of evidence, drug availability, and expert consensus. It directly determines what kind of treatment options will be seen in clinical practice and the priority of adoption of these options.
The CSCO guideline mainly divides the recommendation levels into three levels. Level I recommendation is the standard treatment option that patients should give priority to under current conditions, and represents a mature solution that has proven efficacy and is affordable and accessible to most patients. Level II recommendation indicates a high level of evidence, but slightly poor accessibility, or a slightly lower level of evidence, but a high degree of expert consensus. Level III recommendation indicates a lower level of evidence, but is commonly used in clinical practice or has exploratory value, and is considered acceptable by experts.
In the era of cancer diagnosis and treatment dominated by evidence-based medicine, CSCO guidelines are not only a pocket book for clinicians, but also a touchstone for measuring the value of innovative drugs. Recently, with the readout of Phase II clinical data of Zejing Pharmaceutical's trispecific antibody ZG006 as a monotherapy for patients with refractory advanced small cell lung cancer, we dismantled the diagnosis and treatment path based on CSCO guidelines and tried to analyze the drug's future clinical positioning, market access potential and its path to breakout in market competition.
Lung cancer is a malignant tumor with high morbidity and mortality rates in China and around the world, among which small cell lung cancer accounts for approximately 13%-17% of lung cancers. Although the proportion is low, due to the large overall base of lung cancer, its impact cannot be ignored. The treatment plan for small cell lung cancer is strictly based on the stage at diagnosis. About 30% of patients are in the localized stage, and 70% of the patients are in the extensive stage, that is, the tumor has spread beyond the chest.
The treatment options for limited-stage small cell lung cancer are mainly surgery, chemotherapy and radiotherapy. Immune checkpoint inhibitors combined with chemotherapy are mainly used in the first-line treatment of extensive-stage small cell lung cancer. Although small cell lung cancer is very sensitive to initial treatment, most patients develop relapse and drug resistance after initial treatment.
Existing second-line treatment drugs mainly include topotecan, rubitidine, irinotecan and other chemotherapy drugs, and their efficacy is generally limited. Third-line and above treatment options are even rarer. Only one drug, anlotinib, is recommended by CSCO for third-line and above treatment of small cell lung cancer (category 2A).
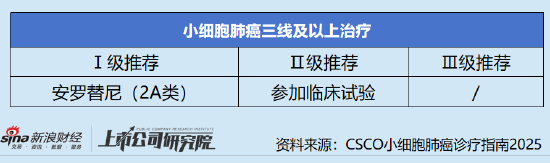
Data show that anlotinib is a new type of small molecule multi-target tyrosine kinase inhibitor independently developed in my country. It can effectively inhibit VEGFR, PDGFR, FGFR, c-Kit and other kinases, and has the effect of anti-tumor angiogenesis and inhibiting tumor growth. The results of the phase II study (ALTER1202) of anlotinib compared with placebo in the third-line and above treatment of small cell lung cancer conducted by Chinese researchers showed that the progression-free survival time of patients after anlotinib treatment reached 4.1 months, which was 3.4 months longer than that of the placebo group; the survival time reached 6.3 months, which was 3.7 months longer than that of the placebo group.
Let’s look at the data performance of Zejing Pharmaceutical ZG006. Clinical data shows that as of September 30, 2025, a total of 60 patients with third-line and above small cell lung cancer were randomly assigned 1:1 to receive 10 mg Q2W or 30 mg Q2W treatment and were included in the analysis. The results based on IRC evaluation showed that the median progression-free survival (mPFS) of the 10 mg Q2W group (30 cases) and the 30 mg Q2W group (30 cases) were 7.03 months and 5.59 months respectively; the median survival time (mOS) of both groups was immature, with 6-month OS rates of 83.2% and 76.7%, and 12-month OS rates of 69.1% and 58.2%, respectively.
Judging from the existing data, ZG006 has demonstrated clinically significant anti-tumor activity in patients with refractory advanced small cell lung cancer, and the data performance is better than existing standard treatments. In addition, the 6-month and 12-month OS rates were 83.2% and 69.1% respectively, which is also a positive signal among the end-of-line treatment population.
However, the current median overall survival data are not yet mature, and the long-term survival benefit still needs to be further confirmed by follow-up data. In the oncology drug evaluation system, OS is still the core endpoint that determines clinical value and guideline recommendation level. If follow-up data can maintain the current trend, ZG006 is expected to become an important supplement for the later-line treatment of small cell lung cancer.
From the competitive landscape, there has been a long-term lack of efficient therapies in the back-line treatment of small cell lung cancer. The current CSCO guidelines only recommend anlotinib (category 2A) in third-line and above treatments, and clinical options are extremely limited. As a trispecific antibody targeting DLL3, ZG006's mechanism involves T cell redirection and immune synapse enhancement. It has a differentiated action path from existing therapies. If its survival benefit can be further verified, it is expected to fill the gap in later-line treatments.
However, if ZG006 wants to maximize its clinical value, it still needs to face many challenges. The first is the risk of success or failure of the key confirmation trial. The current data comes from phase II clinical studies, the sample size is limited, and it is a non-randomized controlled design. Whether subsequent phase III confirmatory studies can replicate the current efficacy signal in a larger sample and randomized controlled population remains to be verified.
Secondly, ZG006 is currently positioned as a third-line treatment and above. Although it can quickly enter clinical unmet needs, from a commercial perspective, the limited patient base for late-line treatment, short treatment cycle, and short drug exposure time limit its market space. If it is unable to expand to more front lines through strategies such as combined treatment or adjuvant treatment in the future, its market ceiling may be significantly restricted.
Taken together, ZG006 has differentiated mechanism advantages and preliminary efficacy signals in the back-line treatment of small cell lung cancer, and has the potential to become a "back-line breakthrough". However, its final clinical positioning and market value will still depend on whether subsequent confirmatory studies can be translated into definite survival benefits, whether safety is controllable, and whether barriers to competition can be formed at the level of medical insurance and guidelines.




